Loading dyes¶
Loading dyes are mixed this samples to pull them down into a well in a gel and to visually track migration during electrophoresis.
When using fluorescently-labelled (Cy3, Cy5) samples, make sure the tracking dyes do not co-migrate with your sample, otherwise you may be unable to tell if a resulting band originates from your sample or the dye.
For native PAGE, do not use dyes that contain SDS as they may separate double-stranded nucleic acids.
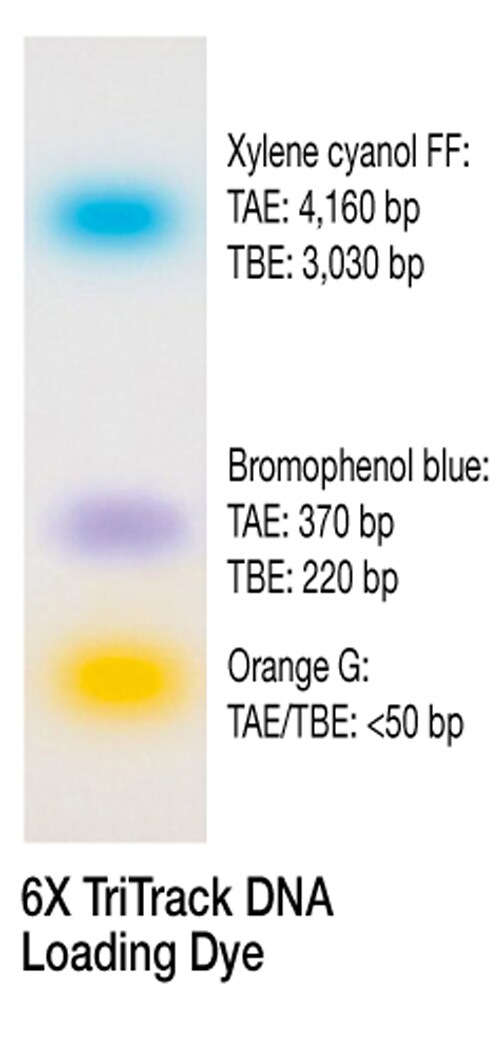
TriTrack¶
- Contents:
- 10 mM Tris-HCl (pH 7.6)
- 0.03 % bromophenol blue
- 0.03 % xylene cyanol FF
- 0.15 % orange G
- 60 % glycerol
- 60 mM EDTA.
- Uses:
- Agarose gel
- Native PAGE
Gel Loading Buffer II¶
- 1-2X solution
- Contents:
- 95% Formamide
- 18 mM EDTA
- 0.025% each of SDS, Xylene Cyanol, and Bromophenol Blue
- Uses:
- Polyacrylamide urea gel (denaturing)
- Non-denaturing agarose gel
- Appearance: Dark blue
Protocol: 1. Mix sample with an equal volume of Gel Loading Buffer II. Vortex briefly. 2. Centrifuge briefly to bring contents of tubes to the bottom. 3. For denaturing PAGE: Heat to 95°C for 5 min to denature any secondary structure. 4. Load directly (while still hot) on the gel.
RNA Loading Dye¶
- 2X
- Contents:
- 47.5% Formamide
- 0.01% SDS
- 0.01% bromophenol blue
- 0.005% xylene cyanol
- 0.5 mM EDTA
- Uses:
- Polyacrylamide urea gel (denaturing)
Protocol: 1. Add sample to an equal volume of RNA Loading Dye, (2X). Mix well. 2. Heat at 65–70°C for 5–10 minutes to denature RNA. 3. While heating the samples, setup the gel box and flush urea out of the wells with running buffer using a large tip. 4. Load samples.